CDC advisory panel to weigh delaying newborn hepatitis B vaccine as health secretary pushes review
Meeting comes after Health Secretary Robert F. Kennedy Jr. replaced ACIP members; panel expected to consider moving the birth dose to age 4 amid debate over infant protection

The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP) is slated to deliberate a proposal to delay the hepatitis B vaccine typically given within 24 hours of birth to children age 4, a move that could alter a long-standing U.S. infant immunization practice.
The proposal is expected to come to a vote at a meeting that will also review COVID-19 vaccine guidance. The possible change follows a broad shakeup of the advisory body: Health Secretary Robert F. Kennedy Jr., who has a long record of questioning vaccine safety, ousted all 17 ACIP members in June and appointed a new slate of advisers ahead of this week’s meeting. "There is going to likely be a discussion about hepatitis B vaccine, very specifically trying to dislodge the birth dose of hepatitis B vaccine and to push it later in life," Demetre Daskalakis, the former director of the CDC’s National Center for Immunization and Respiratory Diseases, told NPR.
Public health officials and medical societies have cautioned that removing or delaying the birth dose could jeopardize progress toward eliminating perinatal transmission of hepatitis B. Hepatitis B is the world’s most prevalent serious liver infection and the leading viral cause of liver cancer. An estimated 296 million people worldwide live with chronic hepatitis B; in the United States, public health officials estimate as many as 2.4 million people may have chronic infections.
In 1991, ACIP recommended universal hepatitis B vaccination for all infants, a policy that led to a steep decline in new cases in the United States. The current routine schedule calls for the first dose within 24 hours of birth, a second dose at 1 to 2 months of age and a third dose between 6 and 18 months. The vaccine prevents about 98% of infections in healthy infants and side effects are generally mild, including soreness at the injection site, redness, headache and fatigue.
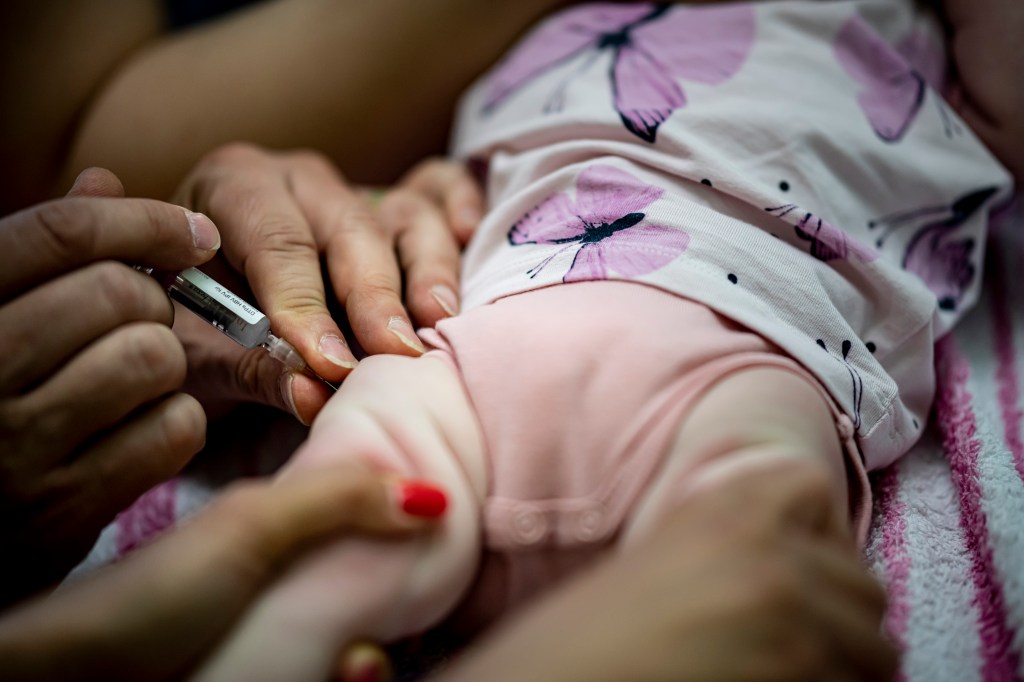
Hepatitis B spreads through contact with infected blood and bodily fluids, most commonly via sexual contact, sharing needles and from mother to child at birth when the mother is infected. U.S. pregnant women are typically screened in the first trimester; if a mother tests positive, clinicians may give antiviral medication in the third trimester and the newborn receives both hepatitis B vaccine and hepatitis B immune globulin (HBIG) within 12 hours of birth to reduce the risk of transmission.
Acute hepatitis B becomes chronic when the immune system does not clear the virus within six months. Children infected at birth are at especially high risk: roughly 90% of infants infected perinatally develop chronic infection, which can progress over decades to liver scarring, failure and cancer if untreated. The CDC reported 17,650 new records of chronic hepatitis B and 1,769 hepatitis B–related deaths in 2023; the agency logged 2,200 acute infections that year but estimated the true number of acute infections to be closer to 14,400 because many cases go undiagnosed.
Medical groups have warned that the birth dose is a critical part of preventing perinatal infection. In June, the American Academy of Pediatrics said the United States was on track to eliminate perinatal hepatitis B and cautioned that removing or delaying the birth dose would "jeopardize this progress." The vaccine’s near-immediate administration at birth is intended to protect infants who could be exposed at delivery before test results for maternal infection are available or before follow-up vaccination can be assured.
There is no cure for hepatitis B; most healthy adults (about 95%) clear acute infection within six months, while those who do not develop chronic disease. Public health experts have pointed to the success of the infant vaccination program in reducing hepatitis B incidence since the early 1990s as a rationale for maintaining the birth-dose strategy.
The ACIP meeting is expected to include presentations on the epidemiology of hepatitis B, vaccine effectiveness and safety data, and modeling of the potential impacts of altering the infant schedule. Any ACIP recommendation would be reviewed by the CDC director before being adopted into federal guidance and could influence state immunization policies and clinical practice. Officials at the meeting have not announced a timeline for any final decision.
The deliberations come as the panel also reviews COVID-19 vaccine recommendations, underscoring the broader reassessment of childhood and adult vaccination policy under the current administration. Public health groups and clinicians will be watching the outcome of the hepatitis B discussion for its implications for infant health and efforts to prevent liver disease across the population.