Eli Lilly to modify Mounjaro KwikPen, reducing leftover 'golden dose' after price surge
Manufacturer says redesigned pen will still deliver four weekly doses; patients who had been extracting an extra dose reacted angrily as wholesale prices more than doubled.
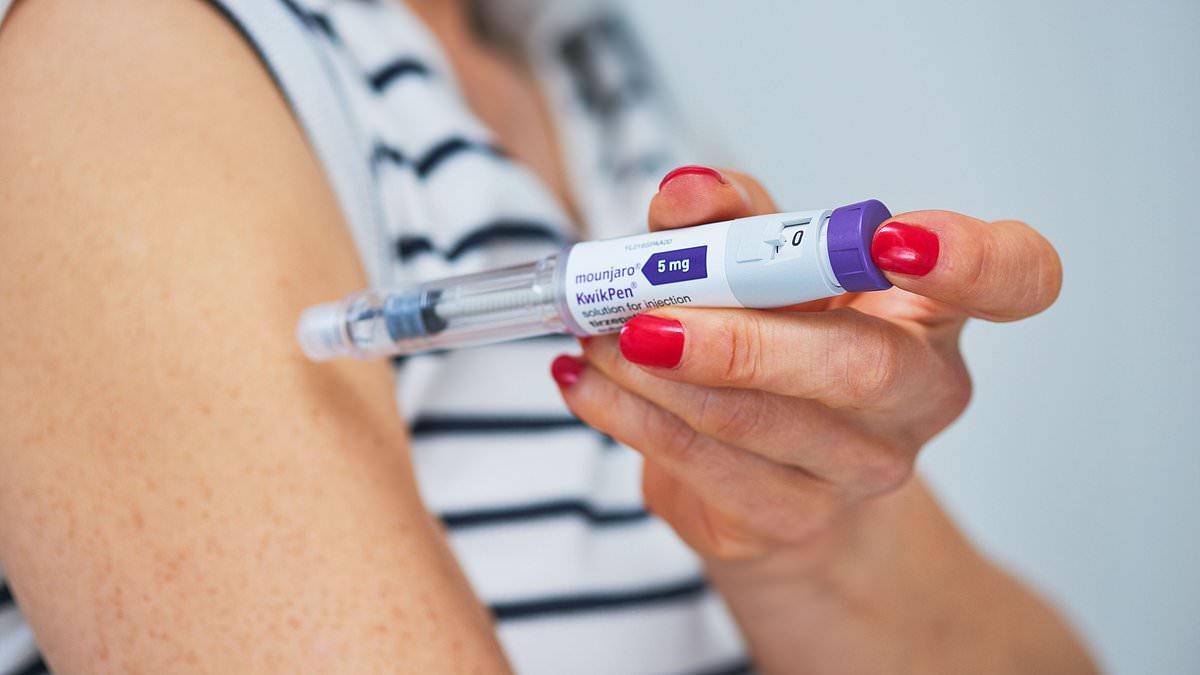
Eli Lilly has confirmed a modified version of its Mounjaro KwikPen that reduces the amount of medicine remaining after the four prescribed weekly doses, a change that will end a practice by some users of extracting an extra so‑called "golden dose" from each pen.
The company said the initial and modified KwikPen formulations both contain the solution needed for priming and delivery of four doses, but that the redesigned pen "has been modified to reduce the amount of leftover medicine that remains in the KwikPen after four doses have been administered," a spokesperson told the Daily Mail. The modified KwikPen has been approved in the U.K., the spokesperson said, but timelines for its availability have not been determined.
Mounjaro pens contain 3 millilitres of solution and are factory‑set to deliver four premeasured weekly doses of 0.6 ml each. Many users on social media have reported that a small amount of liquid remains in the device after the fourth dose and have described methods—using an insulin syringe, needle or manipulating the dose‑setting mechanism—to recover a partial or full additional injection. That practice has been dubbed the "golden dose" and, until now, has allowed some patients to stretch monthly supplies to five weeks.
The change comes as Mounjaro prices have risen sharply. Eli Lilly announced wholesale increases effective Sept. 1 that more than double some monthly prices, with the highest strength cited by the company rising from about £122 to £330. Online pharmacy listings showed wider market movement: Pharmacy2U had a 15 mg pen listed at £314, up from an earlier price of about £180. Mid‑range doses such as the 5 mg pen were reported to increase from roughly £92 to about £180. The price moves and reports of reduced leftover volume prompted some private purchasers to stockpile supplies.
At least half a million patients on the National Health Service and roughly 15 million patients in the United States are now thought to be using GLP‑1 weight‑loss medicines such as Mounjaro, company and industry figures show. U.K. prescribing guidelines reserve these therapies for patients with a body‑mass index (BMI) above 35 with at least one weight‑related condition, or those with a BMI between 30 and 34.9 who meet criteria for referral to specialist weight‑management services.
Clinicians and regulators caution against attempts to extract leftover insulin‑type medications from pens. Health professionals have warned that manipulating pens or transferring contents into other syringes risks contamination, inaccurate dosing and injury, and could compromise sterility or the efficacy of treatment. Those risks were cited by medical commentators on social media and in discussions among users.
Online reaction has been strong. Posts on Reddit and other platforms expressed anger or frustration at the removal of the perceived extra dose and at the price rises. One Reddit user wrote, "Wow. This company are truly the gift that keeps on giving." Another suggested concerns that older and newer pen versions could circulate simultaneously, complicating stockpiling or purchase strategies. Some users indicated they may still attempt to salvage leftover liquid despite warnings.
Eli Lilly said both the original and modified pens provide the volume required for priming and for four full doses, and that the redesign reduces leftover volume while maintaining full delivery of prescribed doses. The company did not provide a public timetable for distribution of the modified device in all markets.
The confirmation of the redesigned KwikPen and the recent price changes underscore tensions between demand for GLP‑1 medicines, their cost, and patient practices outside official dosing guidance. Regulators and clinicians continue to advise patients to follow licensed instructions and to consult prescribers before altering dose delivery or attempting any reuse or transfer of medication.
Sources
- Daily Mail - Latest News - Fresh blow to Mounjaro users as manufacturer Eli Lily reveal KwikPen is REDUCING in size, removing 'golden dose' - after prices increased by 170%
- Daily Mail - Home - Fresh blow to Mounjaro users as manufacturer Eli Lily reveal KwikPen is REDUCING in size, removing 'golden dose' - after prices increased by 170%