Generic semaglutide could slash weight‑loss jab prices in UK
Analysts expect a surge in affordable options as patents expire and rival manufacturers enter the market, potentially widening NHS access to obesity treatments.
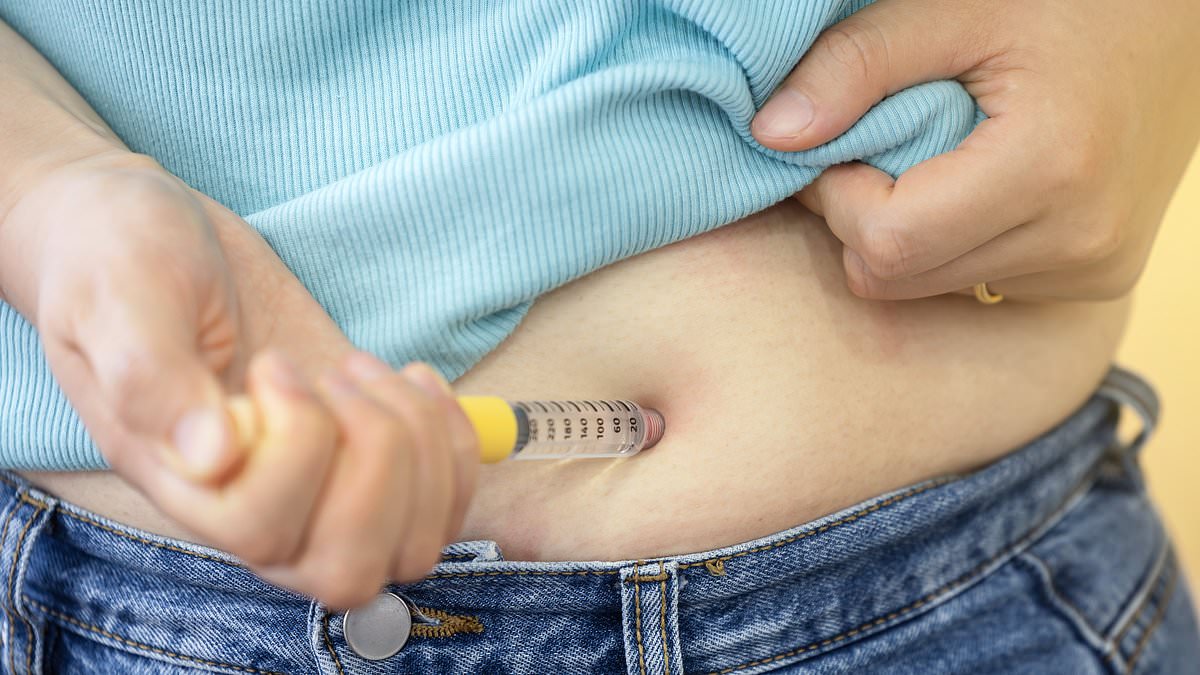
Weight‑loss injections that use semaglutide, the active ingredient in Wegovy and Ozempic, could become markedly cheaper next year as patents begin to expire in large parts of the world, opening the door to generic versions. In the UK, NHS access to these injections remains limited, while many private buyers have paid hundreds of pounds a month through online pharmacies. If early generics roll out in 2026, as some markets anticipate, the price gap between branded and generic semaglutide could widen dramatically and reshape demand for obesity management in Britain.
With patent expiry in countries including India, Canada, China, Brazil and Turkey as soon as next year, dozens of manufacturers are lining up to produce generic semaglutide. Generics typically cost far less than branded versions, and competition can drive price cuts even before patents lapse. Medicines UK notes that generics contain the same active ingredient and are designed to deliver the same therapeutic effects, subject to the same safety and quality standards. The prospect of cheaper versions has prompted a wave of licensing activity as firms prepare to bring versions of semaglutide to market.
In Britain, Novo Nordisk’s UK patent remains in force until 2031, which means the national patent does not yet permit widespread generic imports under current law. Still, experts warn that cheaper, unbranded or imported semaglutide could begin circulating through grey-market channels ahead of official approvals, raising concerns about quality control and the risk of counterfeits. Some patients have already turned to overseas online pharmacies to access the injections while NHS access remains tight; regulators stress that buying prescription medicines outside established channels carries safety risks.
Industry observers say the arrival of generics could be a turning point for public health in the UK. Obesity already costs the NHS an estimated £6.5 billion each year, with more than one million hospital admissions in England linked to the condition annually. Without effective interventions, officials warn that the bill could rise to about £9.7 billion by 2050, straining frontline services. Experts emphasise that generic semaglutide will still need to meet rigorous quality standards, and that price reductions are not guaranteed to be immediate or uniform across every segment of the market. The head of Medicines UK stressed that generics offer a potential long‑term public‑health advantage by enabling broader treatment access, particularly for higher‑risk populations.
Several manufacturers have publicly outlined plans to bring semaglutide to market at lower prices. Adalvo announced it was preparing to offer injections across the full dosing range—from the lowest to the highest approved doses. Swiss generics giant Sandoz also revealed plans to launch unbranded semaglutide next year, projecting discounts of about 60% to 70% compared with branded versions. The European Patent Office recently rejected an attempt by Teva and Galenicum Health to extend Novo Nordisk’s semaglutide patent to 2033, underscoring the growing momentum behind generic competition. Novo Nordisk said patent expiry is a natural part of the lifecycle of a pharmaceutical product and that the company remains committed to innovation and exploring new molecules, combinations and formulations to help people with obesity and diabetes worldwide.
In Britain, the rollout of weight‑loss jabs has been described as creating a two‑tier system, with affording access largely limited to those who can pay. A May report from the Tony Blair Institute for Global Change found obesity rates are about 15% higher in the most deprived areas than in the wealthier segments of society, and it warned that limited NHS access intensifies disparities. It also highlighted concerns about vulnerable patient groups who may not receive appropriate, personalised online support when obtaining injections through private channels. Health chiefs have called for caution over misuse as doctors report rising numbers of patients seeking slimming injections via online chemists, sometimes after misrepresenting weight to obtain prescriptions. Clinicians note that semaglutide injections can help users shed substantial weight on average—about 15 kilograms (around 33 pounds) over roughly 68 weeks—by mimicking natural hormones that suppress appetite.
From a regulatory standpoint, generic semaglutide will be produced under strict manufacturing controls and oversight. Medicines UK says generics are manufactured in plants inspected to Good Manufacturing Practice standards, with quality, safety and efficacy aligned with branded drugs. Britain’s Medicines and Healthcare products Regulatory Agency (MHRA) oversees these processes, while in other major markets, the FDA and the European Medicines Agency perform similar roles. The World Health Organization’s GMP guidelines are widely adopted internationally, helping ensure consistency of quality across borders as competition increases. Analysts expect a healthy level of competition to emerge, potentially enabling the NHS to treat more patients at lower cost, though the lag between patent expiry in other jurisdictions and UK‑specific approvals means price reductions may materialise gradually rather than immediately.
Public health officials caution that the path from price announcements to actual patient access is not automatic. Prescription requirements remain in force, and the risk of counterfeit or unsafe products circulating online persists if patients seek cheaper options outside regulated channels. Officials emphasise the importance of verifying supplier credibility and consulting healthcare professionals before starting any weight‑loss medication. The evolving landscape will depend on how quickly generic suppliers obtain licenses for the UK market, how supply chains adapt to demand, and how clinicians integrate these therapies with existing obesity management programs. As safer, more affordable options potentially widen, the ultimate test will be whether expanded access translates into measurable reductions in obesity‑related illness and hospital admissions across the country.