Trump administration set to tie Tylenol use in pregnancy to autism risk, outline leucovorin research
Administration plans to advise against over-the-counter Tylenol during pregnancy and to explore leucovorin as a potential autism treatment, with HHS chief Kennedy Jr. weighing in on new CDC findings.
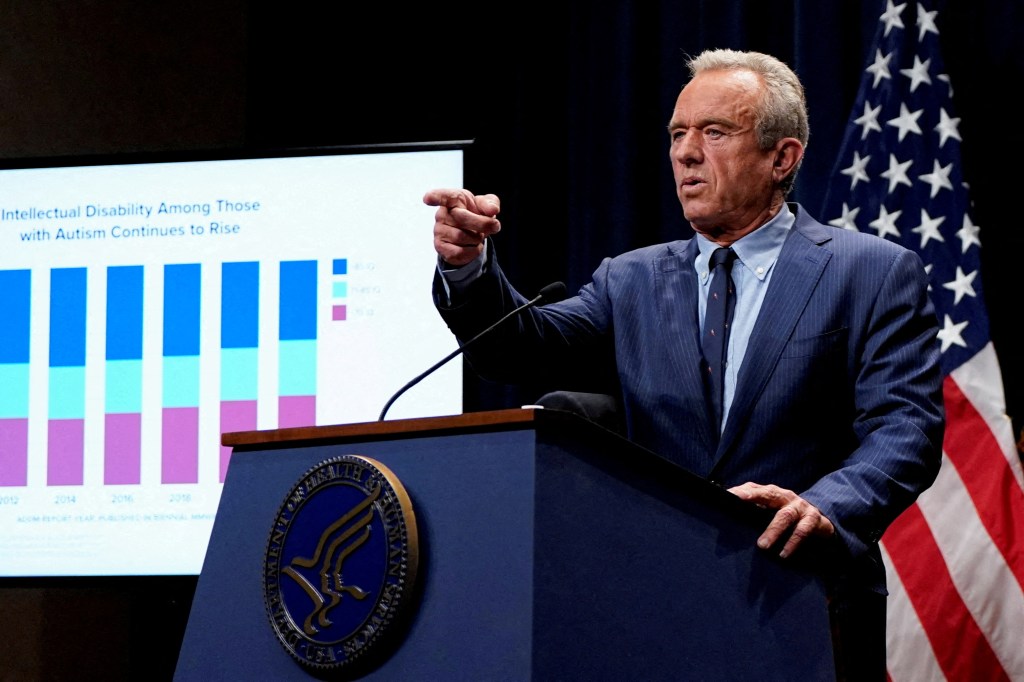
The Trump administration is preparing to announce that using acetaminophen, the active ingredient in Tylenol, during pregnancy could be linked to an increased risk of autism in children, according to multiple people familiar with the matter. The White House also plans to outline a research and policy agenda that would include exploring leucovorin, a drug used to treat certain cancers and anemia, as a potential autism treatment. The formal news conference is scheduled for Monday.
Officials say the administration will advise pregnant women to avoid over-the-counter Tylenol except when a fever is present, arguing that the potential risk warrants caution. The proposal would also detail a broader effort to study autism therapies and prevention strategies, including trials of leucovorin. In conjunction with the briefing, Health and Human Services Secretary Robert F. Kennedy Jr. is expected to discuss the latest findings from the Centers for Disease Control and Prevention's Autism and Developmental Disabilities Monitoring Network. Trump has been previewing a major announcement in recent days, telling supporters at the funeral of conservative influencer Charlie Kirk that he believed a breakthrough on autism was near.
The plan signals a shift in how the administration frames everyday medications and childhood development in public health messaging. While officials say the move is driven by data reviews, the announcement would come as part of a broader effort to shape public expectations about science and health policy from the executive branch. Monday’s event would mark a high-profile assertion of a new line of inquiry that ties common analgesics to developmental outcomes and positions leucovorin as a potential therapeutic avenue.
Leucovorin research is described as exploratory. The drug, which is approved for certain cancer therapies and for treating some anemias, is being studied for possible effects on neurological development in children diagnosed with autism. The administration cautions that any use outside approved indications would require rigorous clinical evaluation and regulatory review, and officials emphasize that no final therapeutic approval is expected in the near term.
The Centers for Disease Control and Prevention’s Autism and Developmental Disabilities Monitoring Network provides surveillance data on autism spectrum disorder across multiple sites. The latest ADDM findings are typically released with implications for public health planning and funding, and the White House says the briefing will reference these data as part of the rationale for pursuing new treatment strategies and risk communication around maternal medication use.
Analysts and medical researchers have long urged caution about drawing causal conclusions from observational data on acetaminophen exposure during pregnancy. The administration’s approach to present a potential risk association alongside new treatment research will likely draw scrutiny from clinicians who stress the need for robust, peer-reviewed evidence before altering widely used pregnancy guidance. Health experts have stressed that assertions about autism risk require careful consideration of confounding factors and the strength of available data, and officials say the Monday briefing will include detailed citations and plan for follow-up studies to clarify any associations.
If implemented, the policy would represent one of the most visible pivots in public health messaging for the administration, linking a common, over-the-counter medication to a complex neurodevelopmental outcome while simultaneously outlining a potential therapeutic pathway. The White House has not released full supporting documents, and schedule details remain subject to change as health officials coordinate with researchers and clinical trial networks. The administration’s team argues that proactive communication, paired with targeted research, could help families make informed decisions during pregnancy while accelerating exploration of treatments.