White House Reposts Tylenol Warning as Trump Ties Pregnancy Use to Autism Concerns
Administration highlights an old Tylenol post as President Trump cites studies linking acetaminophen to autism; FDA planning label changes.
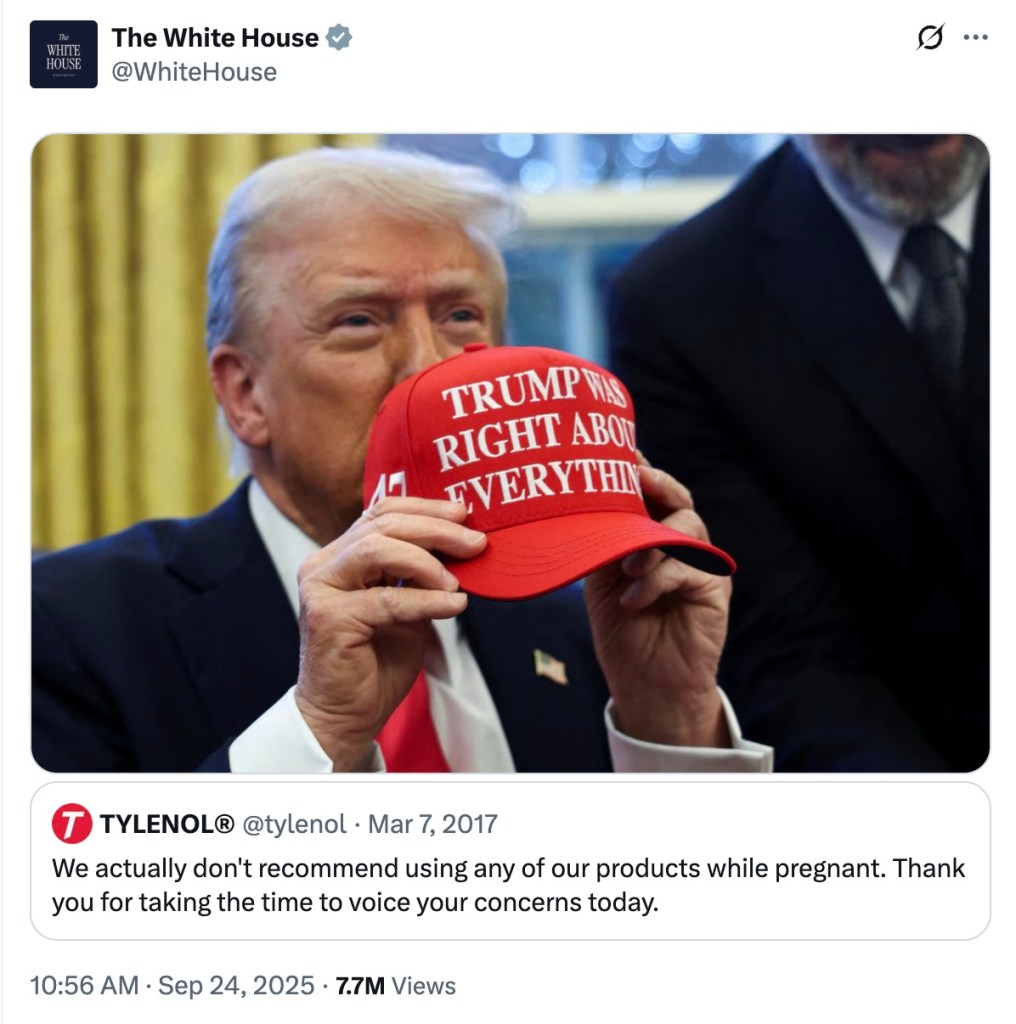
The White House and the Department of Health and Human Services drew attention to a Tylenol post from eight years ago as President Donald Trump argued this week that acetaminophen could be linked to autism, prompting a public reaction on social media.
On Monday, Trump urged pregnant women to avoid Tylenol unless they have a high‑risk fever, saying studies have shown a possible connection between acetaminophen use in pregnancy and autism in children.
The White House’s official X account reposted Tylenol’s 2017 tweet declaring, "We actually don’t recommend using any of our products while pregnant." The post was quickly noticed by political observers and on social media.
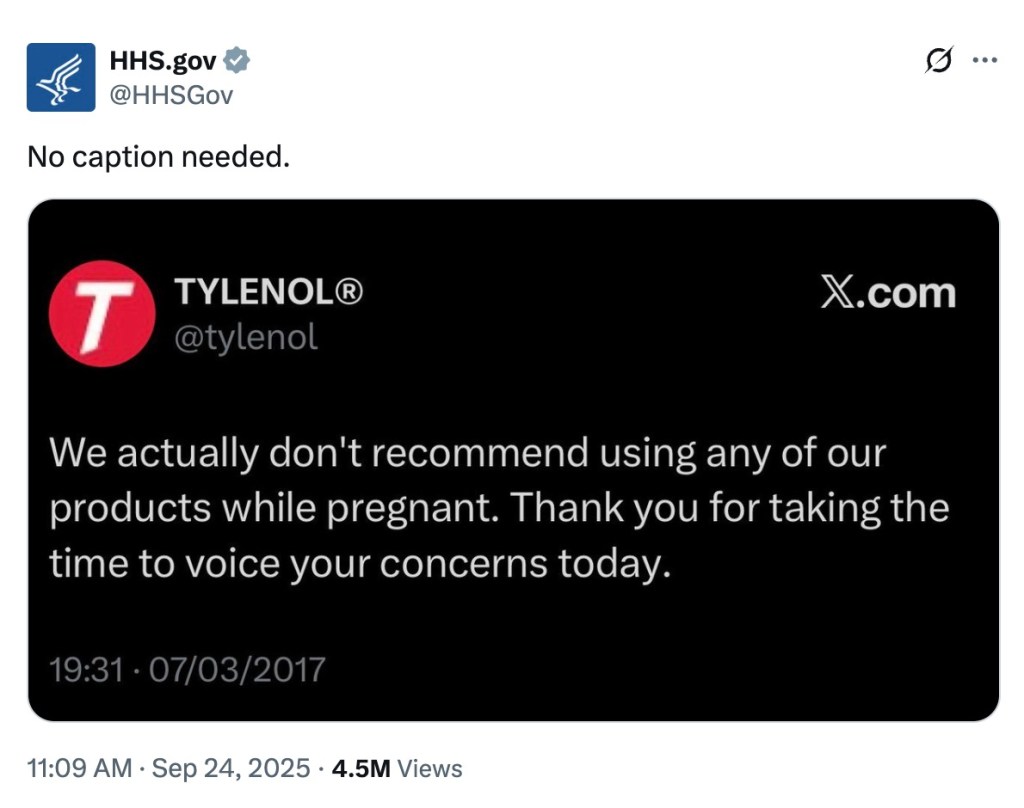
The Department of Health and Human Services also shared a screenshot of the old Tylenol post, a move that amplified the moment alongside the president’s comments.
Tylenol’s parent company, Kenvue, said the repost was taken out of context and stressed that it does not issue recommendations about medication in pregnancy; that guidance remains a healthcare‑provider decision. In a statement, the company said, "This eight-year-old consumer response is incomplete and did not address our full guidance on the safe use of Tylenol which has not changed: Acetaminophen is the safest pain reliever option for pregnant women as needed throughout their entire pregnancy."
Reaction on social media varied. Some Trump supporters used the moment to tout the president’s stance, while others criticized the use of a brand message in policy commentary. One post read, "Would you look at that… Trump was right again." Another commenter wrote, "Liberal women, please take as many Tylenols during pregnancy as possible. Prove Trump wrong, ok?"
The administration said its stance rests on a growing body of research that has examined acetaminophen use during pregnancy and potential links to autism, though medical experts emphasize that the evidence is not conclusive and that acetaminophen is generally considered safe when used as directed. The Food and Drug Administration said it will update the labeling to reflect pregnancy considerations and plans to send a physician advisory on the matter.
FDA labeling changes would accompany a broader push from federal health agencies to clarify risks and ensure clinicians discuss safe use with pregnant patients. Despite the debate, data show that acetaminophen remains the most commonly used over‑the‑counter pain reliever by pregnant women, with more than half reporting use during pregnancy.
Healthcare professionals say decisions about medication during pregnancy should be guided by clinicians, not headlines. The latest episode highlights how political rhetoric can intersect with medical guidance and corporate communications during an ongoing public health conversation.